What are the phases of clinical trials? This journey delves into the intricate process of testing new medical treatments and interventions. From initial safety assessments to large-scale efficacy studies, each phase plays a crucial role in bringing promising therapies to patients. We’ll explore the different types of trials, the participants involved, the ethical considerations, and the meticulous data collection and analysis that underpins the entire process.
Understanding the phases of clinical trials is vital for anyone interested in healthcare innovation. Each phase has specific objectives, participant numbers, and study designs, all carefully orchestrated to ensure the safety and efficacy of new treatments. By examining each stage, we gain a comprehensive understanding of the rigorous process behind bringing new medical breakthroughs to the forefront of patient care.
Introduction to Clinical Trials
Clinical trials are systematic investigations that evaluate the safety and effectiveness of medical interventions, including new drugs, therapies, and medical devices. They are crucial in advancing medical knowledge and improving patient care. These trials play a vital role in transforming hypotheses into evidence-based treatments. They carefully analyze the impact of different interventions on various health conditions.The primary objectives of clinical trials are to assess the efficacy and safety of a specific treatment or procedure compared to a control group.
This comparison allows researchers to determine the treatment’s true benefits and potential risks. A secondary objective is to generate knowledge that can inform future healthcare practices and guidelines.
Different Types of Clinical Trials
Clinical trials are categorized into various phases, each with a specific purpose. Understanding these phases is critical for comprehending the progression of medical research. These phases represent a structured approach to evaluate the safety and efficacy of new treatments.
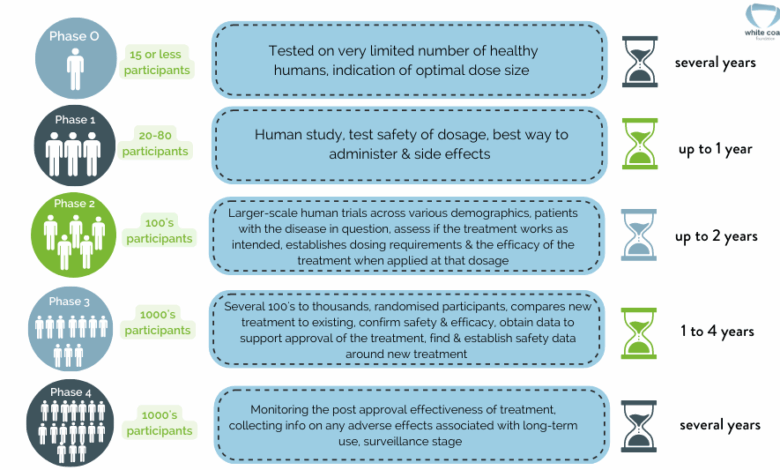
- Phase 1 Trials: These trials are typically small-scale studies involving a small number of healthy volunteers or patients with a specific condition. The primary goal is to evaluate the safety and tolerability of a new treatment. Researchers identify potential side effects, determine the appropriate dosage range, and investigate the best administration method.
- Phase 2 Trials: These trials involve a larger group of patients with the target condition. The primary objective is to further evaluate the treatment’s safety and preliminary effectiveness. Researchers investigate the optimal dose and assess if the treatment produces the desired therapeutic effect in patients.
- Phase 3 Trials: These trials are large-scale studies involving hundreds or thousands of patients. The main goal is to confirm the treatment’s effectiveness and safety in a wider population. Phase 3 trials compare the new treatment to existing treatments or a placebo, providing a rigorous evaluation of its clinical benefit.
- Phase 4 Trials: These trials occur after a treatment has been approved for use. They are conducted to monitor long-term safety, identify rare side effects, and further explore different uses of the treatment in various patient populations. These studies contribute to a comprehensive understanding of the treatment’s effects over time.
General Process of Conducting a Clinical Trial
Clinical trials follow a standardized process, ensuring rigor and integrity in data collection and analysis. These steps are crucial for minimizing bias and maximizing the reliability of the results.
- Protocol Development: A detailed protocol Artikels the trial’s objectives, design, procedures, and data analysis methods. This document serves as a roadmap for the entire trial process.
- Recruitment of Participants: Researchers identify and enroll eligible participants who meet the specified criteria. This process ensures that the study population accurately reflects the target patient group.
- Data Collection and Management: Data on patient outcomes, treatment responses, and adverse events are meticulously collected and managed. This meticulous process ensures data integrity and reliability.
- Data Analysis and Interpretation: Statistical analysis is used to assess the treatment’s efficacy and safety. The findings are critically reviewed and interpreted in the context of the study objectives.
- Dissemination of Results: The findings are published in peer-reviewed journals, shared at medical conferences, and made accessible to the broader scientific community. This dissemination contributes to the advancement of medical knowledge and best practices.
Clinical Trial Types Table
The following table summarizes the different types of clinical trials, their primary purpose, and key characteristics.
| Trial Type | Purpose | Key Characteristics |
|---|---|---|
| Phase 1 | Assess safety and tolerability of a new treatment | Small-scale, healthy volunteers or patients; determines dosage range and administration method |
| Phase 2 | Evaluate preliminary effectiveness and further safety | Larger group of patients; optimal dose and therapeutic effect assessment |
| Phase 3 | Confirm effectiveness and safety in a wider population | Large-scale; compares new treatment to existing treatments or placebo |
| Phase 4 | Monitor long-term safety and explore different uses | Post-approval; investigates rare side effects and broader applications |
Phases of Clinical Trials
Clinical trials are meticulously designed to evaluate the safety and effectiveness of new treatments, interventions, or diagnostic tools. Understanding the different phases of these trials is crucial for grasping the journey a potential therapy takes from initial testing to possible widespread use. Each phase has specific objectives, and their results are vital for determining whether a treatment warrants further development.The progression through these phases is not a straight line.
Sometimes, a treatment shows promise in one phase but not in the next, necessitating adjustments or even abandonment of the study. Or, a trial may need to be paused due to unforeseen issues, highlighting the dynamic nature of clinical research.
Phase 1 Trials
Phase 1 trials are the first step in testing a new treatment in humans. These trials are focused on safety and determining the appropriate dosage. Researchers aim to identify potential side effects and determine how the treatment is absorbed, distributed, and metabolized by the body. The primary objective is not to prove effectiveness, but to establish a safe dose range for future trials.
Clinical trials, crucial for testing new treatments, typically progress through phases. Understanding these phases is important, especially when considering the potential benefits and risks. For example, while there are potentially harmful germs on public transport, like subways, many are actually quite harmless, much like some early-stage clinical trial results. Learning about the phases of clinical trials is like learning about the subway ecosystem, where you need to differentiate the safe from the potentially hazardous; lots of germs on subway most harmless is a helpful comparison.
Ultimately, the phases of clinical trials help us understand the journey of new treatments towards approval.
- Participants: Typically, a small number of healthy volunteers or patients with the specific condition are involved.
- Study Design: Phase 1 trials often employ a dose-escalation design, gradually increasing the dosage to find the maximum tolerated dose (MTD) without causing significant harm.
- Data Collection Methods: Researchers meticulously monitor participants for side effects, blood tests, and other indicators of how the body responds to the intervention. These trials may also involve observing physical examination findings and other clinical markers.
- Interventions: Examples include new cancer drugs, novel vaccines, and new therapies for rare diseases.
Phase 2 Trials
Phase 2 trials build on the safety data from Phase 1 to evaluate the treatment’s effectiveness in patients with the specific condition being studied. These trials are designed to determine if the treatment has a measurable effect and to further refine the dosage.
- Participants: A larger group of patients with the target condition is recruited.
- Study Design: These trials employ various study designs, including randomized controlled trials (RCTs), which help to ensure that participants are as similar as possible to one another. Researchers seek to understand how effective the intervention is in treating the disease.
- Data Collection Methods: Researchers collect data on the treatment’s effectiveness, including measures of the disease’s severity, side effects, and participant responses to the treatment.
- Interventions: For example, evaluating the efficacy of a new chemotherapy regimen for lung cancer or a new drug for treating depression.
Phase 3 Trials
Phase 3 trials are large-scale, randomized, controlled trials that compare the new treatment to an existing standard of care or a placebo. The primary objective is to confirm the treatment’s effectiveness and safety in a larger and more diverse population.
- Participants: The largest number of participants are involved in this phase. The goal is to determine the efficacy and safety in a diverse patient population.
- Study Design: Phase 3 trials often employ randomized controlled trials (RCTs), comparing the new treatment to an existing standard treatment or a placebo. This helps to establish the treatment’s efficacy.
- Data Collection Methods: Extensive data collection is performed to assess the treatment’s effectiveness and safety in a larger and more diverse population. This phase may use a wide range of clinical measurements.
- Interventions: Examples include evaluating a new blood pressure medication, investigating the effectiveness of a new surgical procedure, and comparing different dosages of an existing medication.
Phase 4 Trials
Phase 4 trials are conducted after a treatment has been approved for marketing. These trials focus on monitoring the long-term effects, identifying rare side effects, and exploring different ways to use the treatment.
- Participants: Typically involve a large number of patients who are already using the treatment.
- Study Design: Phase 4 trials often employ observational studies and post-marketing surveillance.
- Data Collection Methods: Researchers monitor patients who have received the treatment over time to collect data on long-term outcomes, side effects, and optimal usage strategies.
- Interventions: Monitoring the long-term effects of a new cholesterol-lowering drug or evaluating different ways to use a new medication in different patient populations.
Comparison of Clinical Trial Phases
| Phase | Participant Numbers | Study Design | Data Collection Methods |
|---|---|---|---|
| 1 | Small | Dose-escalation | Safety monitoring, blood tests |
| 2 | Larger | Controlled or uncontrolled trials | Effectiveness and side effects |
| 3 | Largest | Randomized controlled trials | Extensive data collection |
| 4 | Large | Observational studies | Long-term effects and optimal use |
Participants in Clinical Trials: What Are The Phases Of Clinical Trials
Clinical trials are crucial for advancing medical knowledge and improving patient care. A critical component of these trials is the diverse group of participants who volunteer to contribute to the research process. Understanding the different types of participants, the selection criteria, and their rights is essential for ensuring the ethical and effective conduct of clinical trials.The success of a clinical trial hinges on carefully selecting participants who accurately represent the population the treatment is intended for.
This ensures that the results are generalizable and applicable to a wider patient base. Furthermore, understanding the rights and responsibilities of participants safeguards their well-being and fosters trust in the research process.
Types of Participants
Clinical trials involve various participant roles, each with specific responsibilities. Understanding these roles is critical for the smooth execution of the research. The primary participants are patients, often those with specific diseases or conditions. Other participants include investigators, who conduct the study, and sponsors, who fund and oversee the trial.
Participant Selection Criteria
Rigorous criteria are used to select participants for clinical trials. These criteria ensure that the trial population is homogenous enough to accurately measure the treatment’s effectiveness while still encompassing the diversity of the target population. Inclusion criteria define the characteristics that a participant must possess to be eligible, while exclusion criteria Artikel characteristics that would prevent a participant from participating.
These criteria often include factors like age, gender, medical history, and current health status. For example, a trial for a new cancer drug might include patients with a specific type of cancer but exclude patients with other severe comorbidities.
Rights and Responsibilities of Participants
Participants in clinical trials have a set of rights and responsibilities. Their rights include the right to be fully informed about the trial, the right to refuse to participate or withdraw at any time without penalty, and the right to confidentiality. Participants also have a responsibility to adhere to the trial protocol, attend scheduled appointments, and provide accurate information.
A participant’s informed consent is paramount, ensuring they understand the risks and benefits associated with the study.
Informed Consent
Informed consent is a crucial ethical consideration in clinical trials. It’s a process that ensures participants fully understand the nature of the trial, including the risks and benefits, before agreeing to participate. This ensures the participant is making a voluntary and informed decision about their participation. The process of obtaining informed consent typically involves providing participants with a detailed explanation of the trial, outlining the procedures, potential risks, and benefits.
Participant Roles
| Role | Description |
|---|---|
| Patient/Participant | Individuals enrolled in the trial to receive the treatment or intervention being studied. |
| Investigator | Researchers who conduct the study, manage the participants, and collect data. |
| Sponsor | Organization (often a pharmaceutical company) that funds and oversees the clinical trial. |
| Data Management Team | Responsible for collecting, managing, and analyzing the data from the trial. |
Ethical Considerations in Clinical Trials
Clinical trials, crucial for advancing medical knowledge and treatments, must be conducted with the utmost ethical rigor. Protecting the rights and well-being of participants is paramount. This section delves into the ethical principles guiding these trials, the role of Institutional Review Boards, informed consent procedures, and participant safety measures.
Key Ethical Principles Governing Clinical Trials
Ethical principles form the bedrock of clinical trials. These principles ensure that the trials are conducted in a manner that prioritizes the welfare of participants while advancing scientific knowledge. A fundamental principle is respect for persons, recognizing participants’ autonomy and right to make informed decisions. Beneficence mandates researchers act in the best interests of participants, minimizing potential harms and maximizing benefits.
So, clinical trials have distinct phases, right? From initial testing to large-scale studies, each phase has specific goals. For example, imagine the recent findings about microplastics released into a cup of tea, microplastics released into cup of tea. Understanding the effects of these tiny particles on human health is likely to involve various phases of clinical trials.
These studies would follow strict protocols to ensure safety and effectiveness, just like other medical research.
Justice emphasizes equitable selection of participants and equitable distribution of trial-related burdens and benefits.
Role of Institutional Review Boards (IRBs) in Clinical Trials
Institutional Review Boards (IRBs) play a critical role in safeguarding the ethical conduct of clinical trials. They are independent ethics committees responsible for reviewing research protocols to ensure compliance with ethical standards and regulations. IRBs evaluate the potential risks and benefits of the study, the adequacy of participant protections, and the overall ethical soundness of the research design.
This review process helps prevent harm to participants and maintain public trust in clinical research.
Procedures for Obtaining Informed Consent
Informed consent is a cornerstone of ethical clinical trials. It involves ensuring participants understand the study’s purpose, procedures, risks, and benefits. Researchers are obligated to present information clearly and comprehensively, using language understandable to participants. Participants must comprehend the information provided and voluntarily agree to participate. This process must be entirely voluntary, and participants must be free to withdraw at any time without penalty.
A written consent form, typically signed by both the participant and the researcher, documents the process.
Methods for Protecting the Rights and Well-being of Participants
Protecting the rights and well-being of participants is paramount. Researchers must meticulously monitor participants’ health status throughout the trial, promptly addressing any adverse events. Confidentiality of participant data is essential, and strict protocols must be in place to safeguard sensitive information. Researchers must be transparent about potential risks and benefits. Participants should be provided with mechanisms to address their concerns, and their feedback should be taken seriously.
The principles of minimizing harm and maximizing benefits are critical in ensuring the safety and well-being of trial participants.
Summary of Ethical Guidelines and Regulations
| Ethical Principle | Description | Example |
|---|---|---|
| Respect for Persons | Recognizing the autonomy and rights of participants | Ensuring participants are free to withdraw from the trial without penalty. |
| Beneficence | Acting in the best interests of participants | Minimizing potential risks and maximizing benefits of the intervention. |
| Justice | Equitable selection of participants and distribution of burdens and benefits | Ensuring diverse representation in the trial population. |
| Informed Consent | Providing comprehensive information and obtaining voluntary agreement | Providing participants with a clear and concise explanation of the study protocol. |
| Data Privacy and Confidentiality | Protecting the privacy of participant data | Implementing secure storage and access control mechanisms for participant data. |
Data Collection and Analysis in Clinical Trials
Clinical trials are meticulously designed to gather and analyze data to determine the effectiveness and safety of new treatments or interventions. This process requires a rigorous approach to ensure accurate and reliable results. The careful collection and analysis of data are crucial to ensuring that the findings can be generalized to a larger population. The integrity of the results directly impacts the potential for a successful treatment to be adopted in clinical practice.
Methods of Data Collection
Data collection in clinical trials employs a variety of methods to ensure comprehensive and reliable information. These methods are carefully chosen based on the specific research questions and the characteristics of the participants. Each method has its strengths and weaknesses, and the most appropriate one for a given situation is selected to maximize the accuracy of the data.
- Questionnaires and Surveys: These tools are used to collect self-reported data on demographics, lifestyle factors, symptoms, and patient experiences. They are often used to gather baseline information and track changes over time.
- Medical Records Review: Reviewing medical records provides access to a comprehensive history of the participant’s health, including diagnoses, treatments, and laboratory results. This provides context and potentially reveals factors that might influence the trial results.
- Physical Examinations: Physical examinations are used to assess the participant’s physical condition, identify any changes, and document findings. These examinations may involve measuring vital signs, performing neurological checks, or assessing other relevant physical characteristics.
- Laboratory Tests: Blood tests, urine tests, and other laboratory procedures provide objective measurements of various biological markers. These can be used to monitor treatment effects, assess side effects, and detect potential complications.
- Imaging Studies: X-rays, CT scans, MRIs, and other imaging techniques provide visual representations of internal structures and can be used to assess treatment responses, track disease progression, and monitor for adverse events.
Data Analysis Process
The analysis of data collected from clinical trials is a multifaceted process, requiring specialized statistical techniques to determine the significance of the findings. This process aims to determine if the intervention has the intended effect and if it is safe for use.
- Data Cleaning and Preparation: Raw data is initially checked for errors, inconsistencies, and missing values. This stage is critical because inaccurate or incomplete data can lead to flawed conclusions. Data entry errors, missing values, and outliers are identified and addressed.
- Descriptive Statistics: Descriptive statistics summarize and describe the collected data, providing a general overview of the sample. This involves calculating measures of central tendency (mean, median, mode), dispersion (standard deviation, variance), and frequencies. This helps in understanding the characteristics of the data and identifying patterns.
- Inferential Statistics: Inferential statistics are used to draw conclusions about the population from the sample data. This often involves hypothesis testing, statistical modeling, and regression analysis. This determines whether observed differences or effects are likely due to chance or a real effect of the intervention.
- Statistical Significance and Interpretation: The results are interpreted in light of the statistical significance and clinical relevance. The statistical significance of the results is determined using p-values and confidence intervals. This ensures that the results are robust and can be generalized to a larger population. The clinical significance is assessed to determine the practical importance of the findings.
Types of Data Collected
Clinical trials collect various types of data, categorized broadly as demographics, clinical outcomes, and safety data. Each type provides crucial insights into the intervention’s effectiveness and safety profile.
- Demographics: This includes characteristics like age, gender, ethnicity, and socioeconomic status. These factors are important because they can influence the response to treatment.
- Clinical Outcomes: These are the primary and secondary measures used to evaluate the effectiveness of the intervention. They could include changes in disease severity, symptom improvement, or functional capacity.
- Safety Data: This encompasses information about adverse events, side effects, and any other complications that arise during the trial. This data is crucial for assessing the risks associated with the intervention.
Evaluating Effectiveness and Safety
The data collected and analyzed are used to determine the effectiveness and safety of the intervention. This involves comparing the outcomes of the intervention group to those of a control group (if applicable) or to historical data.
Understanding the phases of clinical trials is crucial for evaluating new treatments. It’s a complex process, but think of it like building a house – each phase is a step. For example, a good lunch can prevent a mid-afternoon slump and keep your energy levels consistent, just like a solid foundation supports a house. Checking out this recipe for a lunch recipe avoid energy crash can help you avoid those afternoon energy crashes.
Ultimately, the different phases of clinical trials help determine if a treatment is safe and effective for widespread use.
- Effectiveness: The data analysis helps determine whether the intervention produces the desired effect. This is often assessed by comparing the rates of improvement, remission, or cure between the intervention and control groups.
- Safety: The analysis of safety data helps identify any potential risks or side effects associated with the intervention. This includes the frequency and severity of adverse events, as well as the relationship between the intervention and any observed complications.
Data Collection and Analysis Flowchart
| Step | Description |
|---|---|
| 1 | Define research questions and data elements |
| 2 | Develop data collection tools and procedures |
| 3 | Recruit and enroll participants |
| 4 | Collect data from participants |
| 5 | Clean and prepare data for analysis |
| 6 | Perform descriptive and inferential analysis |
| 7 | Interpret results and draw conclusions |
| 8 | Disseminate findings |
Regulatory Approval and Oversight

Navigating the complexities of clinical trials requires meticulous adherence to regulatory guidelines. These guidelines, established by governing bodies like the FDA, ensure the safety and efficacy of new treatments before they reach patients. This process involves rigorous evaluation, stringent reporting requirements, and ongoing oversight to maintain public trust and protect patient well-being.Regulatory approval is a multi-stage process, not a simple yes-or-no decision.
It involves a careful review of data from all phases of clinical trials, followed by a thorough evaluation of the drug’s safety profile and efficacy. The goal is to balance the potential benefits of a new treatment with the inherent risks, ensuring that the benefits outweigh the risks for patients.
The FDA’s Role in Clinical Trial Approval
The Food and Drug Administration (FDA) in the US plays a crucial role in regulating the development and approval of new drugs and medical devices. They act as a gatekeeper, ensuring that products are both safe and effective for their intended use. This involves a rigorous assessment of data generated during clinical trials, ensuring compliance with established standards and regulations.
Requirements for Reporting Trial Results
Detailed reporting of clinical trial results is essential for transparency and accountability. This reporting must be comprehensive, including both positive and negative findings. The FDA and other regulatory bodies require specific data formats and content to be included in these reports. This ensures that all relevant information is accessible for thorough review. Failure to adhere to these requirements can delay or even halt the approval process.
Examples of Regulatory Documents Related to Clinical Trials
Several key documents Artikel the regulatory requirements for clinical trials. These include:
- Investigational New Drug (IND) application: This application is submitted to the FDA prior to initiating clinical trials. It details the proposed trial design, methodology, and the drug’s potential benefits and risks.
- Clinical Trial Protocols: Detailed plans for conducting the trial, outlining procedures, patient selection criteria, and data collection methods. These documents are critical for ensuring consistency and comparability across trials.
- Case Report Forms (CRFs): Used to record data collected during the trial. They are standardized forms to maintain consistency and ensure data integrity.
Regulatory Steps in Bringing a Drug to Market
The process of bringing a drug to market is complex and multi-layered. Here’s a table outlining the key regulatory steps:
| Step | Description |
|---|---|
| Pre-clinical Testing | Initial testing on animals to assess safety and efficacy before human trials. |
| IND Application Submission | Submission to the FDA for approval to conduct clinical trials. |
| Clinical Trials (Phases I-III) | Testing on human volunteers to evaluate safety and efficacy. |
| New Drug Application (NDA) Submission | Submission of data from all clinical trials to the FDA for review. |
| FDA Review and Decision | Thorough assessment of the submitted data by the FDA, potentially leading to approval, denial, or requests for further information. |
| Post-Market Surveillance | Ongoing monitoring of the drug’s safety and effectiveness after it’s approved for use. |
Reporting and Dissemination of Results
Clinical trials, meticulously designed and executed, culminate in a crucial phase: the reporting and dissemination of results. This process is paramount for the advancement of medical knowledge and ultimately, patient care. Accurate and transparent reporting ensures that the findings are effectively utilized by the medical community and contribute to evidence-based decision-making.
Methods for Reporting Clinical Trial Results
Clinical trial results are reported using standardized methods to ensure accuracy, reliability, and comparability. These methods often include rigorous statistical analysis, adherence to predefined protocols, and meticulous documentation of all data collected throughout the trial. This systematic approach allows researchers and healthcare professionals to draw meaningful conclusions from the data, reducing the risk of bias and misinterpretation. Furthermore, the transparency in reporting is crucial for evaluating the validity and reliability of the trial results.
Formats for Presenting Clinical Trial Results
Dissemination of clinical trial results occurs through various formats, each serving a specific purpose and audience. Peer-reviewed publications in medical journals are a cornerstone of disseminating research findings. These publications provide detailed analyses and discussion of the trial results, allowing for scrutiny and critique by the scientific community. Conferences and symposiums offer platforms for oral presentations, enabling direct interaction with researchers and healthcare professionals.
These presentations often summarize key findings and highlight areas for future research.
Importance of Transparency in Reporting
Transparency in reporting clinical trial results is essential for building trust and fostering accountability. Full disclosure of all aspects of the trial, including its methodology, participants, funding sources, and limitations, is vital. Transparency allows the scientific community to evaluate the results critically and identify potential biases or limitations. This rigorous scrutiny ultimately enhances the validity and reliability of the research findings.
Open access to trial data and protocols is another key aspect of transparency.
Process of Disseminating Trial Findings, What are the phases of clinical trials
The process of disseminating trial findings involves several stages. First, the trial results are analyzed and prepared for publication. Second, these results are submitted to relevant peer-reviewed journals for publication. Third, presentations are prepared and delivered at conferences to disseminate findings to a broader audience. Finally, the trial results are made available to the public through various channels, ensuring accessibility and facilitating ongoing research.
Channels for Communicating Clinical Trial Results
| Channel | Description | Example |
|---|---|---|
| Peer-reviewed medical journals | Rigorous publication process with expert peer review. | The New England Journal of Medicine, JAMA |
| Medical conferences and symposiums | Oral presentations and posters to disseminate results to a wider audience. | American Medical Association Annual Meeting |
| Online databases | Centralized repositories for clinical trial information and results. | ClinicalTrials.gov |
| News outlets | Public reporting of significant trial findings. | Medical news articles in the press |
| Professional organizations | Dissemination through newsletters and publications for specific medical specialties. | American Heart Association |
Dissemination through these various channels allows for a multifaceted approach, catering to diverse audiences and facilitating a comprehensive understanding of the trial results within the medical community. This diverse approach ensures that the research findings reach a broad audience, enhancing their impact on medical practice.
Challenges and Future Trends

Clinical trials, while crucial for advancing medical knowledge and improving patient care, face numerous hurdles. From ethical considerations to logistical complexities, the path to successful trial execution is often fraught with challenges. Addressing these issues is vital for maintaining the integrity and efficiency of the process. Looking ahead, innovative technologies and approaches are poised to revolutionize clinical trial design and execution, promising a more streamlined and effective system for bringing new treatments to patients.
Challenges in Clinical Trials
The execution of clinical trials is often hampered by a multitude of factors. These include recruitment difficulties, particularly for specific patient populations. High dropout rates during trials can lead to incomplete data sets, compromising the reliability of results. Maintaining data integrity and adhering to strict ethical guidelines throughout the entire trial process is a significant challenge. Financial constraints and regulatory hurdles also pose obstacles to the efficient conduct of trials.
The complexities of international collaborations can further complicate the process, requiring meticulous coordination and oversight.
Future Trends in Clinical Trials
Several promising trends are shaping the future of clinical trials. The adoption of technology, such as wearable sensors and remote monitoring devices, is revolutionizing patient recruitment and data collection. Artificial intelligence (AI) is being increasingly utilized for patient stratification, data analysis, and prediction modeling. This trend has the potential to significantly improve the efficiency and effectiveness of clinical trials.
Furthermore, the development of decentralized trials, allowing for remote participation and data collection, is gaining traction.
Evolving Technologies and Approaches in Clinical Trial Design
Technological advancements are reshaping the landscape of clinical trial design. Real-time data collection using wearable sensors allows for continuous monitoring of patient outcomes, providing a richer and more nuanced understanding of treatment efficacy. The integration of AI algorithms enables faster and more accurate identification of potential treatment candidates and tailoring treatment protocols based on individual patient characteristics. Decentralized trials, which leverage telehealth and remote monitoring, are expanding access to clinical trials for a wider range of patients, particularly those in geographically remote areas.
These evolving approaches are paving the way for more efficient, accessible, and patient-centric clinical trials.
Use of Technology in Streamlining Clinical Trials
Technology plays a pivotal role in streamlining clinical trials. Remote patient monitoring systems, for example, allow for continuous data collection and reduce the need for frequent clinic visits. This not only improves patient convenience but also reduces the overall cost and time associated with trials. Electronic data capture (EDC) systems facilitate secure and efficient data management, minimizing errors and enhancing data integrity.
These technological advancements are making clinical trials more efficient, accessible, and reliable.
Potential Areas for Future Research in Clinical Trials
Future research in clinical trials should focus on several key areas. Developing more sophisticated AI algorithms for patient stratification and risk prediction is crucial for optimizing trial design and improving treatment outcomes. Further investigation into the ethical implications of AI in clinical trials is essential to ensure responsible and equitable use. Improving recruitment strategies for diverse patient populations and fostering greater international collaborations will increase the generalizability and impact of trial results.
Addressing the challenges of data privacy and security in the context of remote monitoring and decentralized trials is another important area for future research. These areas hold the potential to transform the field of clinical trials, leading to more efficient, accessible, and impactful research.
Final Conclusion
In conclusion, the phases of clinical trials represent a meticulously designed process, crucial for evaluating the safety and efficacy of new medical treatments. From initial testing to large-scale studies, each phase contributes to the development of life-saving therapies. This intricate system of testing ensures that interventions are thoroughly evaluated before they reach patients, reflecting the commitment to responsible medical advancement.
By understanding the entire process, from ethical considerations to data analysis, we gain a deeper appreciation for the dedication and rigorous standards inherent in clinical research.