Breakthrough T1D Advances Understanding of Disease-Modifying Therapies Amidst Complex Clinical Trial Results
The pursuit of transformative treatments for Type 1 Diabetes (T1D) has taken a significant turn with recent updates from Breakthrough T1D, a leading organization dedicated to eradicating the disease. While the organization champions a multi-pronged strategy to develop disease-modifying therapies (DMTs) capable of halting, slowing, or even reversing T1D, the latest interim analysis from Diamyd Medical’s Phase 3 DIAGNODE-3 clinical trial has presented unexpected challenges. This development, though not meeting its primary endpoint, underscores the intricate nature of immune therapies and reinforces Breakthrough T1D’s commitment to rigorous scientific inquiry and sustained investment in the T1D research pipeline.
Breakthrough T1D’s overarching mission is to fundamentally alter the trajectory of Type 1 Diabetes for all affected individuals, including those yet to be diagnosed. This ambitious goal encompasses the development of DMTs that aim to prevent the onset of T1D, halt its progression, or restore the body’s ability to produce insulin. The organization’s strategic approach integrates two primary therapeutic avenues: immune therapies designed to rebalance the body’s autoimmune response and beta cell therapies focused on preserving or regenerating insulin-producing cells.
DIAGNODE-3 Trial: An Interim Analysis Reveals Unexpected Outcomes
On March 27, Diamyd Medical, a company at the forefront of T1D therapeutic development, announced the results of an interim analysis of its DIAGNODE-3 Phase 3 clinical trial. This pivotal trial, partially funded by Breakthrough T1D, was designed to evaluate the efficacy of Diamyd (retogatein or rhGAD65) in preserving endogenous insulin production, as measured by C-peptide levels, in individuals with recent-onset T1D. The analysis focused on the first 174 participants out of a planned total of 321.
Phase 3 clinical trials represent a critical juncture in drug development, serving as the final hurdle before regulatory agencies like the U.S. Food and Drug Administration (FDA) can consider a therapy for approval. These large-scale studies are designed to confirm the safety and efficacy of a treatment in a diverse patient population.
In a surprising turn of events, the interim analysis of the DIAGNODE-3 trial indicated that Diamyd did not demonstrate a statistically significant preservation of insulin production (C-peptide) in the evaluated cohort. This finding contrasted with promising data observed in earlier phases of the clinical trial, highlighting the complexities inherent in developing immune-modulating therapies for T1D. The unexpected outcome serves as a stark reminder of the scientific hurdles that must be overcome in the quest for effective DMTs.
Breakthrough T1D’s Perspective: Learning from Every Study
In the wake of these results, Breakthrough T1D has issued a measured and forward-looking perspective, emphasizing the invaluable lessons learned from all clinical trials, regardless of their immediate outcomes. The organization views Phase 3 trials, even those that do not meet their primary endpoints, as essential components of the scientific process. The data generated, even from unexpected results, provides critical insights that can refine future research, inform the design of subsequent trials, and accelerate the development of more targeted and effective therapies.
Dr. Josh Vieth, Senior Director of Research at Breakthrough T1D, articulated the organization’s stance: "Progress in type 1 diabetes research and treatment has always required persistence and learning from every study. These results reinforce that immune interventions in type 1 diabetes are complex and highly context dependent. It’s imperative that we take these results seriously, understand what they mean, and use them to make strategic decisions about what’s next for retogatein and other disease-modifying therapies. There remains an unmet need for safe, effective therapies that rebalance the immune system in a targeted way, and Diamyd Medical is a pioneering approach to precision medicine for type 1 diabetes in clinical applications."
This statement underscores Breakthrough T1D’s unwavering commitment to advancing T1D research. It acknowledges the inherent challenges in modulating the immune system within the complex biological landscape of T1D and highlights the importance of a strategic, data-driven approach to therapeutic development. The emphasis on "precision medicine" suggests a growing understanding that future T1D therapies may need to be tailored to specific patient profiles or stages of the disease.
The Road Ahead: Data Analysis and Future Directions

Breakthrough T1D is actively engaged with the Diamyd Medical team to meticulously analyze the data from the DIAGNODE-3 trial. The primary objective of this collaborative effort is to understand the reasons behind the divergence of these interim results from previous findings. Such deep-dive analyses are crucial for unraveling the complexities of immune responses in T1D and for identifying potential factors that may have influenced the trial’s outcome.
Diamyd Medical has indicated its intention to continue core operational activities, including manufacturing, during this period of intensive data review. The company will then make informed decisions regarding its strategic path forward. One significant possibility being explored is the potential to test Diamyd in earlier stages of T1D (Stage 1 or Stage 2). This approach is particularly noteworthy as Diamyd previously received "Fast Track" designation from the FDA for earlier stages of the disease. The FDA’s Fast Track program is designed to expedite the development and review of drugs intended to treat serious conditions and fill unmet medical needs, potentially accelerating the path to market for therapies showing promise in specific patient populations.
Breakthrough T1D’s Comprehensive Strategy for Disease-Modifying Therapy Development
The pursuit of disease-modifying therapies for T1D is not a new endeavor for Breakthrough T1D. The organization has been a consistent and significant funder of DMT research for decades. The landmark approval of Tzield (teplizumab-mzwv) in 2022, the first DMT for T1D, marked a watershed moment for the T1D community and validated Breakthrough T1D’s long-term vision and investment strategy.
Breakthrough T1D’s strategy for DMT development is multifaceted, aiming to address both the autoimmune attack on beta cells and support the intrinsic function of these vital cells. A key component of this strategy is "tolerization," a process that involves re-educating the immune system to cease its attack on beta cells without necessitating broad immunosuppression, which can carry significant side effects.
Currently, Breakthrough T1D is actively funding a diverse portfolio of DMT research initiatives. This includes supporting companies like GentiBio in their efforts to test engineered regulatory T cells (Tregs), a type of immune cell crucial for maintaining immune tolerance. Furthermore, the organization is investing in trials focused on repurposing existing drugs for T1D treatment. This includes investigations into the potential of drugs such as DFMO (eflornithine), Bimekizumab, Denosumab, and ATG (anti-thymocyte globulin) to offer new therapeutic avenues for individuals living with T1D.
The Landscape of T1D Research and the Importance of Diversified Investment
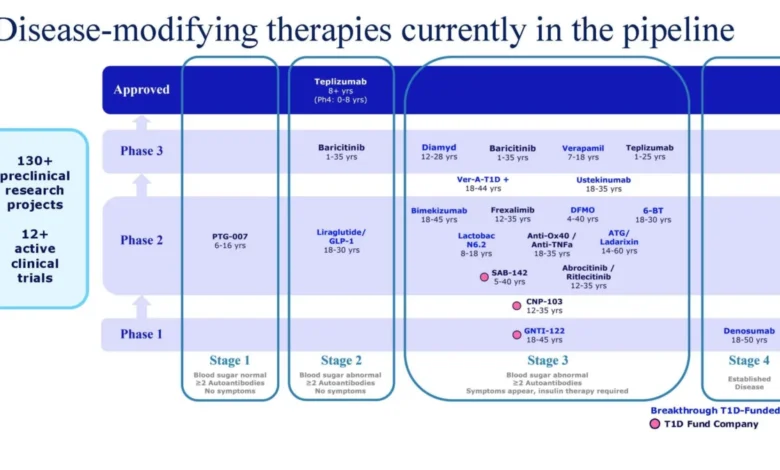
The DIAGNODE-3 trial results, while a setback for that specific program, do not diminish the broader progress being made in T1D research. The accompanying image, referenced in the original article, visually represents the robust pipeline of DMTs currently in various stages of clinical development. It illustrates the breadth of research efforts, spanning Phase 1, 2, and 3 trials, and targeting different stages of T1D, from early onset (Stage 1) to more advanced disease. This visual representation underscores the significant investment and scientific effort being poured into finding transformative treatments.
The complexity of T1D, an autoimmune disease where the body’s own immune system mistakenly destroys insulin-producing beta cells in the pancreas, necessitates a diverse and persistent research approach. Immune therapies aim to interrupt this autoimmune cascade, while beta cell therapies focus on either protecting existing beta cells or promoting the regeneration of new ones. The success of any single therapy can depend on a multitude of factors, including the stage of disease at which it is administered, the specific genetic and immunological profile of the patient, and the precise mechanism of action of the drug.
The approval of Tzield, which delays the onset of Stage 3 T1D in at-risk individuals by a median of two years, has provided crucial proof-of-concept for DMTs. However, the goal remains to develop therapies that can achieve more profound and lasting effects, such as halting or reversing the disease entirely. Breakthrough T1D’s continued funding of a wide array of research projects, including those exploring novel immune modulation techniques and repurposing established drugs, reflects a strategic commitment to exploring multiple avenues for achieving this ultimate objective.
The journey towards a cure or effective long-term management of T1D is a marathon, not a sprint. Each clinical trial, whether successful or not, contributes vital pieces to the scientific puzzle. The commitment of organizations like Breakthrough T1D, coupled with the dedication of researchers and the resilience of individuals living with T1D, fuels the ongoing pursuit of a future where this chronic condition is no longer a life-altering diagnosis. The lessons learned from the DIAGNODE-3 trial will undoubtedly inform and strengthen future endeavors, bringing the T1D community closer to its ultimate goal.








