Understanding the Nuances of Type 1 Diabetes: From Onset to the Elusive Honeymoon Phase and Future Research Frontiers
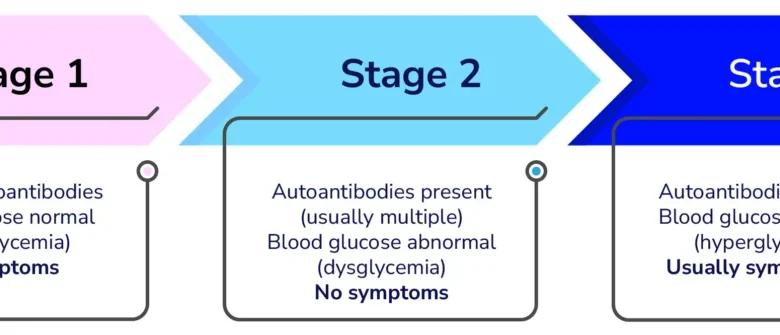
Type 1 diabetes (T1D) is a chronic autoimmune condition where the body’s immune system mistakenly identifies insulin-producing beta cells in the pancreas as foreign invaders and systematically destroys them. This relentless attack is not an instantaneous event but rather a progressive erosion of these vital cells over an extended period. As more beta cells succumb to the immune assault, the body’s capacity to produce insulin, a crucial hormone responsible for regulating blood glucose levels by allowing cells to absorb sugar for energy, diminishes. This decline in insulin production leads to a cascade of metabolic disruptions, characterized by abnormal blood sugar fluctuations. Ultimately, when a critical mass of beta cells has been destroyed, insulin production plummets to insufficient levels, triggering the onset of overt symptoms that typically lead to a diagnosis.
The insidious nature of T1D onset means that individuals often remain asymptomatic for years, even as the autoimmune process unfolds. The pancreas, a versatile organ located behind the stomach, plays a dual role in digestion and hormone production. Within its Islets of Langerhans reside specialized cells, including the beta cells, which are the sole source of insulin. The immune system’s misguided targeting of these cells initiates a complex biological drama. Initially, the body may compensate for the gradual loss of beta cells by making existing cells work harder. However, this compensation has its limits.
The Diagnostic Crossroads: When Symptoms Emerge
The clinical manifestation of T1D typically occurs when the destruction of beta cells has progressed to a point where the body can no longer produce enough insulin to maintain safe blood glucose levels. Common symptoms include increased thirst (polydipsia), frequent urination (polyuria), unexplained weight loss, extreme hunger (polyphagia), fatigue, and blurred vision. In some cases, particularly if the onset is rapid, individuals may present with diabetic ketoacidosis (DKA), a life-threatening condition characterized by high blood sugar and the presence of ketones in the urine. DKA requires immediate medical attention and hospitalization.
The Elusive Honeymoon Phase: A Temporary Respite
Following a diagnosis of T1D, some individuals, though not all, may experience a period known as the "honeymoon phase." This phase is characterized by a temporary improvement in glycemic control, where the remaining beta cells in the pancreas continue to produce some insulin. This residual insulin production acts as a buffer, helping to moderate blood glucose levels and reduce the immediate need for exogenous insulin therapy. During this period, individuals may require lower doses of insulin, and their blood sugar levels might be more stable and easier to manage, approaching target ranges with less difficulty.
The presence and duration of the honeymoon phase are highly individual and influenced by several factors. Research suggests that individuals who are not diagnosed with DKA, who present with lower HbA1c (glycated hemoglobin) levels at diagnosis, and who are children aged five years or older are more likely to experience this temporary remission. HbA1c is a measure of average blood glucose levels over the preceding two to three months, and lower levels at diagnosis often indicate that the autoimmune destruction of beta cells has not progressed as extensively.
Factors Influencing the Honeymoon Phase:
- Severity of Initial Beta Cell Destruction: The extent to which beta cells have been destroyed at the time of diagnosis is a primary determinant of whether a honeymoon phase will occur and how long it might last. A less advanced stage of destruction leaves more functional beta cells capable of insulin production.
- Age at Diagnosis: While not an absolute rule, younger children may have a more aggressive autoimmune process, potentially leading to a shorter or absent honeymoon phase. Conversely, older children and adults might have a more gradual progression.
- Presence of Diabetic Ketoacidosis (DKA): Diagnosis with DKA often signifies a more advanced stage of beta cell loss, making the honeymoon phase less probable.
- Genetic Predisposition and Immune System Response: Individual genetic makeup and the specific nature of the immune system’s response to the beta cells can influence the trajectory of the disease and the likelihood of experiencing a honeymoon phase.
The Temporal Nature of the Honeymoon Phase
It is crucial to emphasize that the honeymoon phase is inherently temporary. While its duration varies significantly from person to person, it typically lasts for several months and can extend up to one to two years in some cases. During this period, the remaining beta cells, while producing insulin, are still under attack by the immune system. Their functional capacity will inevitably decline as the autoimmune process continues unabated.
The perceived ease of management during the honeymoon phase can sometimes lead to a false sense of security. However, healthcare providers consistently stress the importance of adherence to the prescribed insulin regimen and regular monitoring of blood glucose levels, even during this seemingly more stable period. This proactive approach ensures that individuals are prepared for the eventual loss of endogenous insulin production and the subsequent increase in insulin requirements.

The Quest for Extending the Honeymoon Phase: Research at the Forefront
The temporary nature of the honeymoon phase presents a significant challenge in T1D management. The prospect of extending this period, or even halting the autoimmune destruction of beta cells altogether, is a major focus of ongoing research. Currently, there are no FDA-approved therapies specifically designed to extend the honeymoon phase. However, significant strides are being made in the development of disease-modifying therapies that aim to slow, halt, prevent, or potentially even reverse the onset of T1D.
Breakthrough T1D, formerly the Juvenile Diabetes Research Foundation (JDRF), has been a pivotal funder of T1D research, investing substantial resources—over $273 million in the past decade—into these crucial areas. Their commitment has propelled numerous promising therapies into various stages of clinical trials.
One such therapy generating considerable interest is baricitinib, a Janus kinase (JAK) inhibitor. JAK inhibitors interfere with signaling pathways that are critical for the function of both immune cells and beta cells. By modulating these pathways, baricitinib holds the potential to dampen the autoimmune attack on the pancreas. In October 2025, Eli Lilly initiated a Phase 3 clinical trial to investigate baricitinib’s efficacy in preserving beta cell function in individuals newly diagnosed with T1D. The ultimate goal of this trial is to determine if the drug can extend the honeymoon phase. Breakthrough T1D’s prior funding of an earlier clinical trial involving baricitinib was instrumental in paving the way for this larger, pivotal study.
The implications of successfully extending the honeymoon phase are profound. It could significantly reduce the burden of daily insulin injections and continuous glucose monitoring for individuals with T1D, leading to improved quality of life and potentially mitigating the long-term complications associated with chronic hyperglycemia.
Beyond the Honeymoon: The Inevitable Progression and Ongoing Management
As the autoimmune process relentlessly progresses, the remaining beta cells are eventually destroyed, leading to a state of absolute insulin deficiency. This marks the end of the honeymoon phase and necessitates lifelong, comprehensive insulin replacement therapy. The daily insulin requirements will gradually increase, and individuals may find it more challenging to maintain stable blood glucose levels.
This transition requires close collaboration with a multidisciplinary T1D care team, which typically includes endocrinologists, certified diabetes educators, dietitians, and mental health professionals. These professionals play a vital role in guiding individuals through the complexities of T1D management, which involves:
- Insulin Therapy: This includes various types of insulin (basal, bolus), delivery methods (syringes, pens, insulin pumps), and strategies for dosing based on food intake, physical activity, and blood glucose readings.
- Blood Glucose Monitoring: Frequent monitoring using a glucose meter or a continuous glucose monitoring (CGM) system is essential to track blood sugar levels and inform treatment decisions.
- Nutrition Management: A balanced diet that accounts for carbohydrate intake is crucial for effective blood glucose control.
- Physical Activity: Regular exercise can improve insulin sensitivity and overall health, but it requires careful planning to prevent hypoglycemia or hyperglycemia.
- Psychosocial Support: Living with a chronic condition like T1D can be emotionally taxing. Access to support groups and mental health professionals is important for coping and well-being.
The Broader Impact and Future Outlook
The ongoing research into T1D is not solely focused on extending the honeymoon phase but also on developing strategies for primary prevention—identifying individuals at high risk and intervening before the autoimmune destruction begins. Advances in immunology and genetics are paving the way for potential preventative measures, offering hope for a future where T1D can be intercepted at its earliest stages.
The journey of a T1D diagnosis is a complex one, marked by an intricate interplay of the immune system, cellular destruction, and metabolic regulation. While the honeymoon phase offers a temporary reprieve, it underscores the critical need for continued research and development of therapies that can fundamentally alter the course of this disease. The dedication of researchers, the commitment of organizations like Breakthrough T1D, and the resilience of individuals living with T1D are collectively driving progress towards a future where T1D is no longer a lifelong burden.
The implications of breakthroughs in T1D research extend beyond improved management of the disease. They have the potential to revolutionize our understanding of autoimmune disorders in general, offering insights that could benefit patients with other conditions like rheumatoid arthritis, lupus, and multiple sclerosis. The pursuit of a cure or highly effective treatments for T1D is a testament to the power of scientific inquiry and the unwavering hope for a healthier future. As clinical trials continue and new therapeutic avenues are explored, the landscape of T1D management is poised for significant transformation, offering a brighter outlook for millions worldwide affected by this challenging condition.








